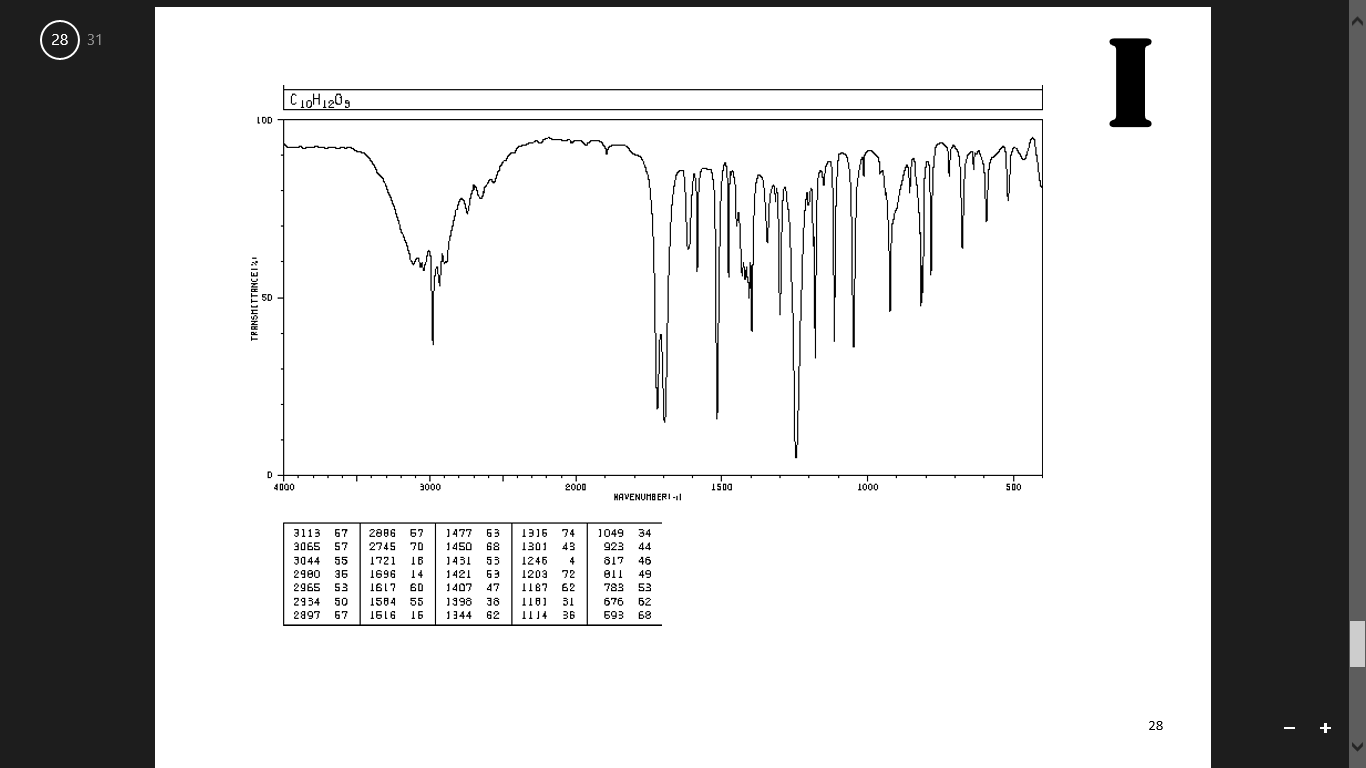 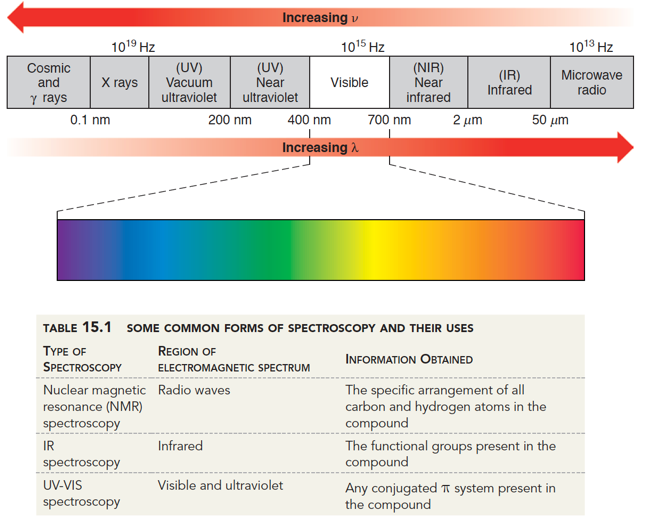
The ν CO modes of 3,5-dioxoindolizidine were found at 1765 cm −1 (five-membered ring) and 1667 cm −1 (six-membered ring) respectively 〈64CB1548〉. IR spectra of indolizidines are extremely useful for conformational assignments 〈71CR109〉, as shown in Section 3.08.2.4.2. The low N N stretching frequency at 2140 cm −1 in diazonium salts of indolizines has been taken as evidence of extensive delocalization (PhN 2 +BF 4 −, 2296 cm −1) 〈67CC424〉. Some routinely recorded spectra have been reported but little effort has gone into correlating the structure with the spectra of these substances. Otherwise little attention has been given to IR spectra of indolizines. A distinction was made between the most characteristic modes of pentagonal and hexagonal rings 〈80SA(A)85〉. All 42 molecular fundamentals were assigned. The IR and Raman spectra of liquid and crystalline indolizine and deuterated derivatives were studied at 200–4000 cm −1. Flitsch, in Comprehensive Heterocyclic Chemistry, 1984 3.08.2.3.3 IR spectra Solute spectra subsequently obtained by DR IR analysis are similar to those obtained by KBr pellet transmittance measurements. Figure 3 illustrates the basic principle. This causes separated mixture components to move to the edge of the TLC plate where they are collected on IR transparent KCl powder. Solute transfer is accomplished by subjecting the developed TLC plate to a second solvent that is applied orthogonally to the direction of the chromatographic mobile-phase flow. To avoid stationary and mobile-phase interferences, solute transfer methods can be used to transport separated mixture components from a TLC plate to an infrared transparent substrate. IR spectra of TLC–IR solutes obtained by DR IR and PA IR exhibit band intensity distortions in spectral regions where stationary and mobile phases absorb and may not resemble those obtained for the same substances by KBr pellet transmittance measurements. White, in Encyclopedia of Spectroscopy and Spectrometry (Third Edition), 2017 Solute Transfer TLC–IR C–O stretch, glycoproteinsĬO–O–C symmetric stretch: phospholipids, cholesterol estersĬ–O stretch, glycoproteins, carbohydrates PO − 2symmetric stretch: lipids, nucleic acids. PO − 2asymmetric stretch: phospholipids, nucleic acids amide III (C–N stretch) of collagenĬO–O–C asymmetric stretch: phospholipids, cholesterol esters ‘Breathing’ vibration of tyrosine ring (C–C/C=C stretching)ĬH 2bend: lipids, proteins, cholesterol estersĬH 3asymmetric bend: lipids, proteins, cholesterol estersĬOO −symmetric stretch: amino acid side chains, fatty acidsĬH 3symmetric bend: lipids, proteins, cholesterol esters Olefinic =CH stretch: lipids, cholesterol estersĬH 3asymmetric stretch: lipids, proteins, carbohydrates, nucleic acidsĬH 2asymmetric stretch: lipids, proteins, carbohydrates, nucleic acidsĬH 3symmetric stretch: lipids, proteins, carbohydrates, nucleic acidsĬH 2symmetric stretch: lipids, proteins, carbohydrates, nucleic acidsĮster C=O stretch: lipids, cholesterol estersĪmide I (amide C=O stretch): proteins, α-helicesĪmide I (amide C=O stretch): proteins, β-sheet In fact, for O–S interactions, a correlation has been found between IR and X-ray crystallographic data 〈71CC1352〉.Īmide B (N–H bend first overtone): proteins Supporting this point of view is the fact that if X-ray structure determinations are not available, the IR spectra are a good proof for some O–S interaction or bonding 〈71AHC(13)161, p. In this chapter we have preferred the oxadithiapentalene names because of the similarities over the whole family of polyheterapentalenes. 220, 72BSF1385, 73BSF3339〉.Īs the experimental S O bond distances given in Section 4.38.2.2 are relatively long, these compounds have been often named as α-(1,2-dithiol-3-ylidene) ketones in the Z configuration. Owing to the fact that the ‘carbonyl’ frequency falls in the region of C C absorption, the identification of the C O stretching vibration has sometimes been established by isotopic substitution with 18O 〈71AHC(13)161, p. The prominent feature of the IR spectra of these compounds is the absence of the usual carbonyl band in the 1620–1720 cm −1 range and the presence of two or more strong bands in the 1500–1610 cm −1 range, a fact considered as the result of a short distance O–S interaction. 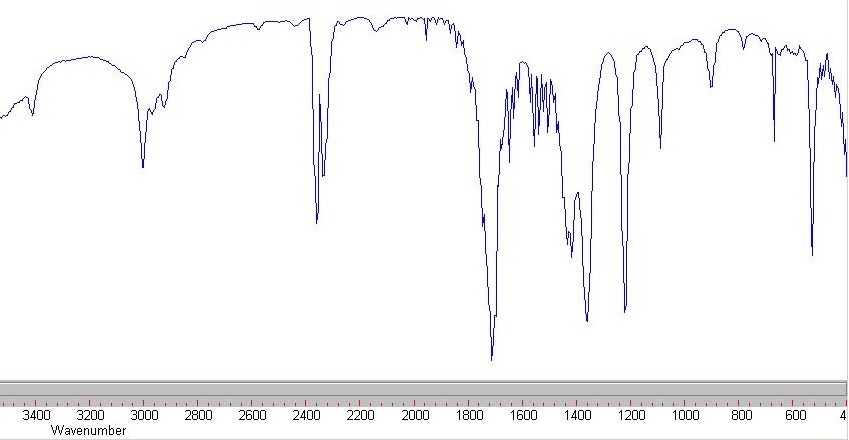
IR spectra have played an important rôle in the structural study of the class of compounds described in this chapter as 1-oxa-6,6aλ 4-dithiapentalenes and often named in the literature as α-(1,2-dithiol-3-ylidene) ketones. Lozac’h, in Comprehensive Heterocyclic Chemistry, 1984 4.38.2.5 IR Spectroscopy
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
